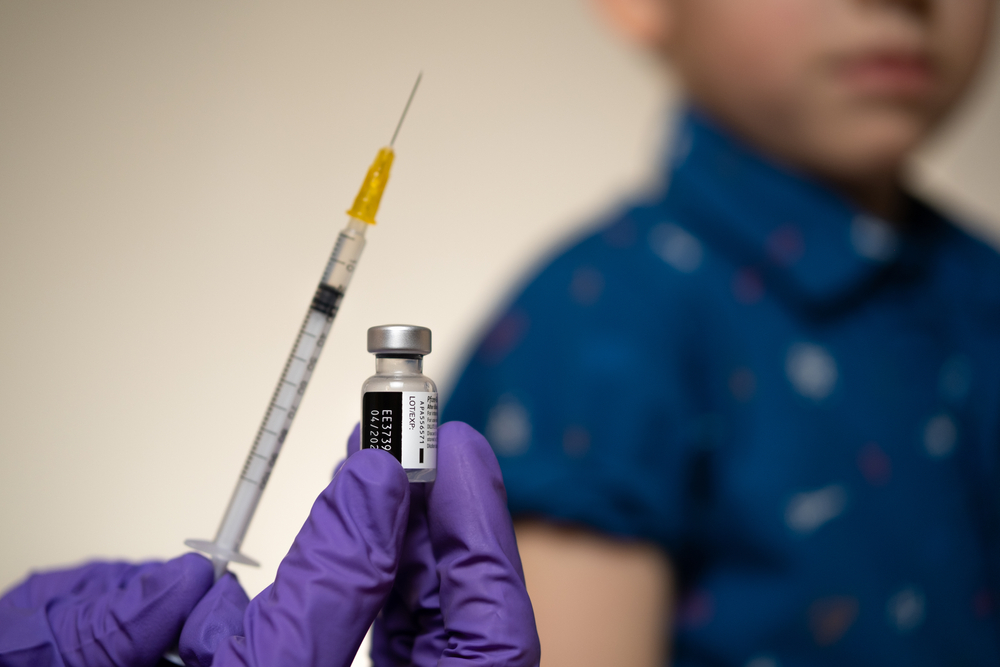
The US Food and Drug Administration is this week expected to approve the distribution of the Pfizer-BioNTech COVID-19 vaccine for children aged 12-15.
Pfizer asked the regulator early last month to authorise the vaccine for children as young as 12. The shot is currently available for anyone aged 16 and over.
Chief Medical Officer Dr. John Lee, speaking on Radio Cayman’s ‘For the Record’ on Monday morning, 3 May, said he understood that the FDA was likely to give the go-ahead this week to administer the vaccine for those over 12.
However, even if the FDA approves the use of the vaccine for 12-15 year olds, this will not automatically mean schoolchildren in Cayman can immediately be vaccinated, Lee said.
“There is a technical problem where we may not be allowed to give it until we get approval from the UK because we are being donated the vaccine from the Foreign, Commonwealth and Development Office, so there is a legal agreement there that we would need to make sure the details are ironed out,” he said.
He added that he expected that by the end of this year, it was likely children as young as 7 could be vaccinated.
In March, Pfizer reported that results of a trial on children within the 12-15 age range “demonstrated 100% efficacy and robust antibody responses, exceeding those recorded earlier in vaccinated participants aged 16 to 25 years old, and was well tolerated”.
In the trial, which involved 2,260 adolescents aged 12 to 15 in the US, 18 cases of COVID-19 were observed in the placebo group versus none in the vaccinated group.
In a statement on 31 March about the trial, Albert Bourla, chairman and chief executive officer of Pfizer, said, “We share the urgency to expand the authorization of our vaccine to use in younger populations and are encouraged by the clinical trial data from adolescents between the ages of 12 and 15.”
BioNTech CEO Ugur Sahin, in the same statement, said, “Across the globe, we are longing for a normal life. This is especially true for our children. The initial results we have seen in the adolescent studies suggest that children are particularly well protected by vaccination, which is very encouraging given the trends we have seen in recent weeks regarding the spread of the B.1.1.7 UK variant. It is very important to enable them to get back to everyday school life and to meet friends and family while protecting them and their loved ones.”
Dr. Hazel Brown, chief nursing officer at the Health Services Authority, who appeared alongside Lee on ‘For the Record’, said people who are continuing to put off getting the vaccine may run out of time to get it once a vaccination drive for the 12-15 age group begins.
She said medical resources, including the vaccines and the staff who administer them, would likely be funnelled towards inoculating this younger portion of the population – similar to how resources were used when the focus was on vaccinating the over-60s population.
“When we start the 12-15 year olds, our focus will shift because we can’t do all the groups at the same time,” she said.
She added, “I’m really pleading with those persons who are 16 and above to come forward and get vaccinated and help us to use our resources.”
As of Friday, 30 April, 65,991 doses of the Pfizer-BioNTech vaccine had been administered in Cayman, with 35,774 people, or 55% of islands’ estimated population of 65,000, receiving at least one dose of the Pfizer-BioNTech vaccine, and 46% having completed the two-dose course.
The rate by which people have been coming forward to be vaccinated has been slowing down in recent weeks.
Cayman’s supply of vaccines will expire at the end of June, and no more are scheduled to arrive on island. Lee said this does not necessarily mean that Cayman won’t get additional supplies in the future, but he urged anyone who is not yet vaccinated to ensure they get their first shot by 9 June at the latest.
Lee invited people who may have suggestions for venues or occasions at which large numbers of people can be vaccinated to put their proposals to the Flu Clinic, which can be contacted on 1-800-534-8600, 947-3077 or [email protected].
Related Videos